CASE REPORT | https://doi.org/10.5005/jp-journals-10018-1337 |
Ruptured Mucinous Cystadenoma Pancreas: A Case Report and Review of Literature
1-4Department of Surgical Gastroenterology, SIMS, Chennai, Tamil Nadu, India
Address reprint requests to: Gautham Krishnamurthy, Department of Surgical Gastroenterology, SIMS, Chennai, Tamil Nadu, India, Phone: +91 1722576565, e-mail: k.gautham@gmail.com
How to cite this article: Krishnamurthy G, Ganesan S, Ramas J, et al. Ruptured Mucinous Cystadenoma Pancreas: A Case Report and Review of Literature. Euroasian J Hepato-Gastroenterol 2021;11(1):45–48.
Source of support: Nil
Conflict of interest: None
ABSTRACT
Mucinous cystic neoplasm (MCN) of the pancreas is a rare neoplasm affecting the elderly women. They vary in their clinical presentation and biological behavior. Spontaneous rupture of MCN is very rare and only eight cases have been reported so far in the English literature. We report a case of a young woman presenting with abdominal pain following spontaneous contained rupture of MCN managed with surgical resection.
Keywords: Cystic neoplasm of pancreas, Distal pancreatectomy, Mucinous cystadenoma, Pancreas, Rupture.
INTRODUCTION
Mucinous cystic neoplasm (MCN) of the pancreas is a rare neoplasm affecting the elderly women.1 They encompass a spectrum of biological behavior from benign cystadenoma to malignant cystadenocarcinomas. They are usually detected incidentally or during evaluation of the vague abdominal symptoms. Spontaneous rupture of MCN is very rare and only eight cases have been reported so far in the English literature. We report a case of a young woman presenting with abdominal pain following spontaneous contained rupture of MCN managed with surgical resection.
CASE DESCRIPTION
A 39-year-old woman presented to the surgical outpatient department with complaints of diffuse abdominal pain for 2 months. There was no other contributory history. Clinical examination was unremarkable except for fullness in the left hypochondrium. Contrast-enhanced computed tomography showed a large peripherally enhancing collection measuring 9.2 × 3.1 cm posteroinferior to proximal body of the stomach. Another peripherally enhancing collection measuring 4.7 × 3.8 cm was noted posteroinferior to the antropyloric region of stomach communicating with the previous collection. The collections were in close relation to the body and the tail of pancreas. Wall of the collection measured 2 to 3 mm in thickness with mild enhancement suggesting mild peritonitis. Endoscopic ultrasound confirmed the collection with aspiration showing mucoid material with no atypical cells on cytology. Fluid analysis showed elevated amylase (1,52,400 IU/L) and lipase (2,10,400 IU/L) with normal carcinoembryonic antigen (CEA) (3.59 ng/mL) and CA19-9 (87.27 U/mL). Magnetic resonance cholangiopancreatography (MRCP) confirmed the suspicion of pancreatic duct communication with an associated proximal duct dilatation (Fig. 1).
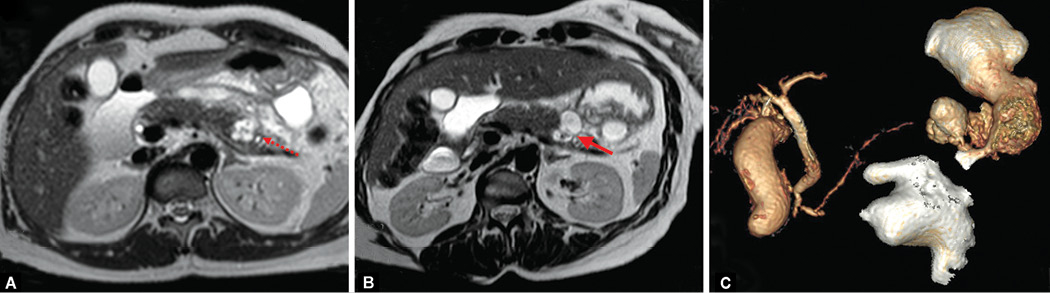
Figs. 1A to C: Magnetic resonance imaging. (A) Axial T2-weighted image showing T2 hyperintense lesion in the pancreatic tail (*) with proximal ductal dilatation (dotted red arrow). (B) Reformatted T2-weighted image showing the communication between the lesion and the collection (solid red arrow). (C) Reconstructed volume-rendered image showing the dilated and the relation of the collection
In view of mucinous aspirate and pancreatic ductal communication, distal pancreatectomy was planned. Intraoperatively, serous fluid of 300 mL in the lesser sac was found tracking along the superior border of the pancreas from body to tail and abutting the transverse mesocolon (Fig. 2). Pancreatic duct disruption was noted at the level of body with mucinous material around the duct (Fig. 2). Distal pancreatectomy with splenectomy was done. The patient had an uneventful postoperative period. Histopathological examination was consistent with the ruptured benign epithelial cystic lesion with ovarian type stroma arising from distal pancreas suggestive of the mucinous cystadenoma (Fig. 3). Twelve regional lymph nodes with reactive changes were also noted. Patient is doing fine at 6 months of follow-up.
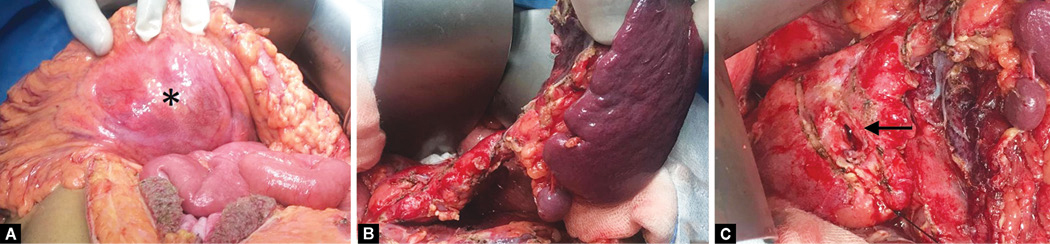
Figs. 2A to C: Intraoperative image. (A) Collection bulging through the transverse colon (*). (B) After complete mobilization of the pancreatic tail and the spleen. (C) Opening of the fistula between the lesion and the pancreatic duct (solid black arrow)
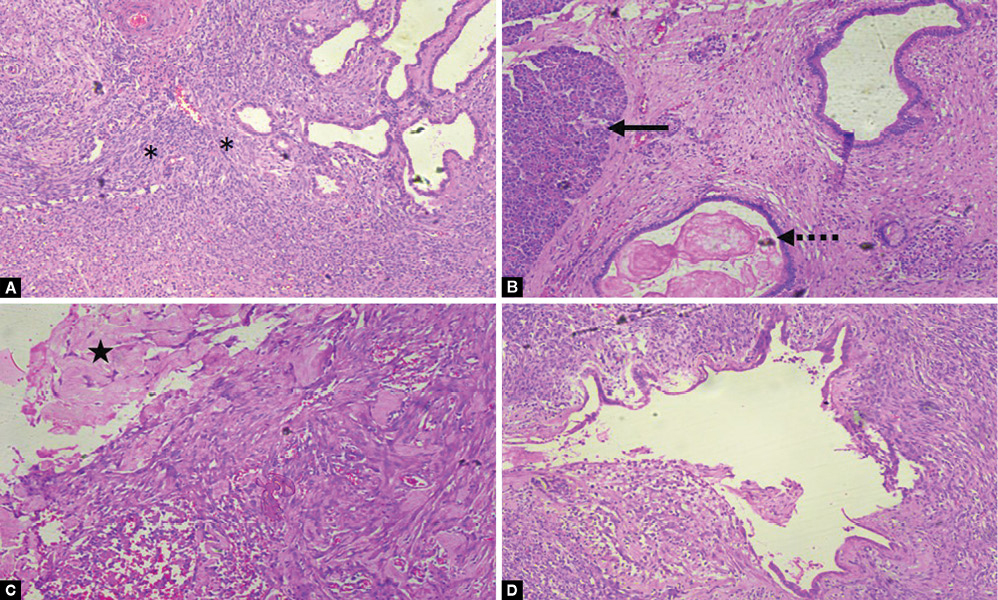
Figs. 3A to D: Photomicrograph of H&E of resected specimen. (A) High-power field (40×) showing presence of ovarian stroma (*). (B) High-power field (40×) showing the presence of adjacent pancreatic acinar tissue (black solid arrow) and dilated pancreatic duct with inspissated mucinous material (dotted black arrow). (C) High-power field (100×) showing corpora albicans. (D) High-power field (40×) showing dilated pancreatic duct
DISCUSSION
MCN of the pancreas represent 1% of pancreatic neoplasms affecting predominately women during the fifth and sixth decades.2 The tumor commonly affects the body and tail of the pancreas.3 De novo mucinous cystadenocarcinoma and malignant transformation of cystadenoma have been described.2 Given their insidious growth and predisposition for affecting the body and tail of the pancreas, patients are often asymptomatic or minimally affected.4 Significant symptoms such as jaundice, back pain, and weight loss may indicate invasive cystadenocarcinoma.4
Apart from malignant transformation, MCNs have very few complications. Spontaneous rupture is one such complication reported. Eight cases of spontaneous rupture of MCN have been reported in the English literature. Table 1 depicts the summary of these cases. The initial case reports were in pregnant women, suggesting a possible stimulation of estrogen or progesterone receptors found on the neoplasms by hormonal changes during pregnancy leading to a rapid increase in size and rupture. However, similar ruptures have been found in nonpregnant women and men. It is also interesting that the histology of the ruptured tumors range from cystadenoma to borderline tumors to even mucinous cystadenocarcinoma. Thus, the exact cause for such a complication is thus difficult to explain. Though the rupture is usually intraperitoneal, even retroperitoneal rupture has been reported.3
| Author | Year | Age | Gender | Presentation | Imaging | Surgery | Final HPR | Follow-up |
|---|---|---|---|---|---|---|---|---|
| Smithers et al.5 | 1996 | 33 | Female | Acute left iliac fossa pain radiating to left shoulder | — | Distal pancreatectomy + splenectomy after cystogastrostomy | Mucinous cystadenocarcinoma | — |
| Ozden et al.2 | 2007 | 32 | Pregnant female | Acute upper abdominal pain | — | Cystectomy with spleen preservation | Well-differentiated mucinous cystadenocarcinoma | Gemcitabine for 6 months. DFS 12 months |
| Bergenfeldt et al.4 | 2007 | 42 | Female | Vague abdominal pain with increasing abdominal distention | USG—massive ascites with large cystic lesion in body of pancreas | Distal pancreatectomy with splenectomy with colectomy | Borderline mucinous cystic neoplasm | DFS—19 months |
| Biswas et al.6 | 2007 | 59 | Female | Acute abdominal pain with loss of consciousness | Hemoperitoneum and a large cystic lesion involving the body and tail of the pancreas | Distal pancreatectomy with splenectomy | Mucinous cystadenoma | — |
| Naganuma et al.7 | 2011 | 32 | Pregnant female | Acute abdomen | Rapidly growing mucinous cystic neoplasm in pancreatic head | Emergency cesarean section with pancreatoduodenectomy | Mucinous cystadenocarcinoma | Four cycles of gemcitabine—local recurrence at 7 months surgically excised. Received additional 16 courses of GEM. DFS—3 years |
| Imoto et al.8 | 2013 | 69 | Female | Abdominal pain | CT—multilocular cystic lesion of the pancreas with solid components with dilated PD. ERCP showed leakage of contrast medium from the main pancreatic duct into the peritoneal cavity | Distal pancreatectomy with splenectomy | Mucinous cystadenocarcinoma | DFS—2 months |
| Woo et al.9 | 2016 | 72 | Male | Epigastric pain for 2 weeks followed by acute exacerbation | CT—irregular high attenuation within the cystic lesion with wall defect. Fluid noted in bilateral paracolic gutter and pelvic cavity | Splenic artery ligation and hematoma evacuation | Frozen biopsy of the nodules of peritoneum, mesentery of the small and large bowel—mucinous cystadenocarcinoma | Adjuvant chemotherapy with gemcitabine and erlotinib. Expired after 189 days of operation due to aspiration pneumonia |
| Haddad et al.3 | 2018 | 30 | — | Acute epigastric pain | MRI—cystic mass at the expense of the body of the pancreas. T2- and T1-weighted imaging showed hypersignal and hyposignal respectively with regular wall enhanced after gadolinium injection. The anterior component had lobulated contours and contained an enhanced septum. A communication between the posterior cystic wall and the retroperitoneum was observed | Distal pancreatectomy with splenectomy | Pancreatic mucinous cystadenoma with low-grade dysplasia | DFS—3 years |
| Our case | 2018 | 39 | Female | Vague abdominal pain | CT and MRCP—Lesser sac collection with pancreatic duct communication | Distal pancreatectomy with splenectomy | Mucinous cystadenoma | DFS – 3 months |
Our index patient was unique in many ways. While she was younger than the usual age of presentation, the subacute nature of the presentation of the contained rupture was not seen in the other cases reported. The imaging and the fluid analysis such as high amylase and normal CEA were favoring pseudocyst. However, the lack of clinical symptom suggestive of acute or chronic pancreatitis and mucinous aspirate suggested otherwise. Communication with pancreatic duct is known to occur in 7% of mucinous cystadenoma explaining the high amylase value and imaging of ductal communication.1 The presence of ovarian stroma differentiates it from the intraductal papillary mucinous neoplasm.10
Emergency surgical intervention was performed in all these cases except one that was incidentally detected during endoscopic retrograde cholangiopancreatogram.8 Excision of the tumor with curative intent has been the principle line of management. In a case of bleeding ruptured MCN, only splenic artery ligation was done by Woo et al.9 in view of biopsy-proven peritoneal disease.
The impact of spontaneous rupture of MCN on overall survival is difficult to ascertain, given the rarity of the complication. Though concerns have been raised about possible tumor spillage during the intervention performed for obtaining tissue or fluid, its impact on survival is not known. We believe that given the good survival shown by other reports, ruptured MCN, especially cystadenoma and borderline tumors, should not be equated to peritoneal involvement, and management should be radical excision with curative intent wherever feasible. In patients with ruptured malignant MCN, adjuvant therapy should be considered to improve overall survival.
CONCLUSION
Spontaneous rupture of MCN is a very rare complication. Preoperative imaging plays a crucial role in identification and planning surgery. Radical excision with curative intent, especially nonmalignant MCN, should be attempted.
CLINICAL SIGNIFICANCE
Spontaneous rupture of cystic neoplasm of pancreas is an uncommon complication. Though more commonly reported in intraductal papillary mucinous neoplasm, clinicians should be aware that the MCNs can also rupture. Moreover, the outcome following rupture depends on the malignant potential of the MCN. Benign and borderline lesions require aggressive surgery to achieve complete cytoreduction and are associated with good overall survival. Malignant lesions have poor outcomes despite adjuvant chemotherapy.
REFERENCES
1. Marques S, Carmo J, Bispo M. Pancreatic mucinous cystadenoma mimicking intraductal papillary mucinous neoplasm. GE Port J Gastroenterol 2018;25(4):201–202. DOI: 10.1159/000480706.
2. Ozden S, Haliloglu B, Ilter E, et al. An extremely rare cause of acute abdomen in pregnancy: ruptured pancreatic mucinous cystadenocarcinoma. Pancreas 2007;34(4):474–476. DOI: 10.1097/mpa.0b013e31803799ee.
3. Haddad A, Sebai A, Rhaiem R, et al. Pancreatic mucinous cystadenoma doubly complicated by acute pancreatitis and retroperitoneal rupture. J Visc Surg 2018;156(1):72–74. DOI: 10.1016/j.jviscsurg.2018.08.011.4.
4. Bergenfeldt M, Poulsen IM, Hendel HW, et al. Pancreatic ascites due to rupture of a mucinous cystic neoplasm. Acta Oncol 2008;47(5):978–981. DOI: 10.1080/02841860701666097.
5. Smithers BM, Welch C, Goodall P. Cystadenocarcinoma of the pancreas presenting in pregnancy. Br J Surg 1986;73(7):591. DOI: 10.1002/bjs.1800730727.
6. Biswas A, Bhattacharya S. Spontaneous rupture of mucinous cystadenoma of the pancreas. Grand Rounds 2006;7:1–4. DOI: 10.1102/1470-5206.2007.0002.
7. Naganuma S, Honda K, Noriki S, et al. Ruptured mucinous cystic neoplasm with an associated invasive carcinoma of pancreatic head in a pregnant woman: report of a case and review of literature. Pathol Int 2011;61(1):28–33. DOI: 10.1111/j.1440-1827.2010.02609.x.
8. Imoto A, Masuda D, Kurisu Y, et al. A case of mucinous cystadenocarcinoma of the pancreas with spontaneous rupture diagnosed by endoscopic retrograde pancreatography. Endoscopy 2013;45 Suppl 2 UCTN:E36–E37. DOI: 10.1055/s-0032-1325888.
9. Woo Y, Kim YD, Jeong WJ, et al. Spontaneous rupture of pancreatic mucinous cystadenocarcinoma: a case report and review of literature. Korean J Pancreas Biliary Tract 2016;21(1):24–28. DOI: 10.15279/kpba.2016.21.1.24.
10. Brugge WR. Diagnosis and management of cystic lesions of the pancreas. J Gastrointest Oncol 2015;6(4):375–388. DOI: 10.3978/j.issn.2078-6891.2015.057.
________________________
© The Author(s). 2021 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and non-commercial reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.